Spine Product Portfolio
All Joline Kyphoplasty products can be purchased individually or as part of the Joline Kyphoplasty Smart Kit — an all-in-one procedural solution containing every device required for a streamlined kyphoplasty workflow.
Kyphoplasty Product Portfolio

US9101
Vertebra Access Device Trocar Tip with Trocar Tip Access Needle

US9102
Guide Pin, blunt

US9201
Vertebra Introducer Device Introducer Cannula & Stylet

US9202
Vertebra Introducer Working Cannula

US9213
RapidIntro Bevel Tip Introducer Cannula & Stylet
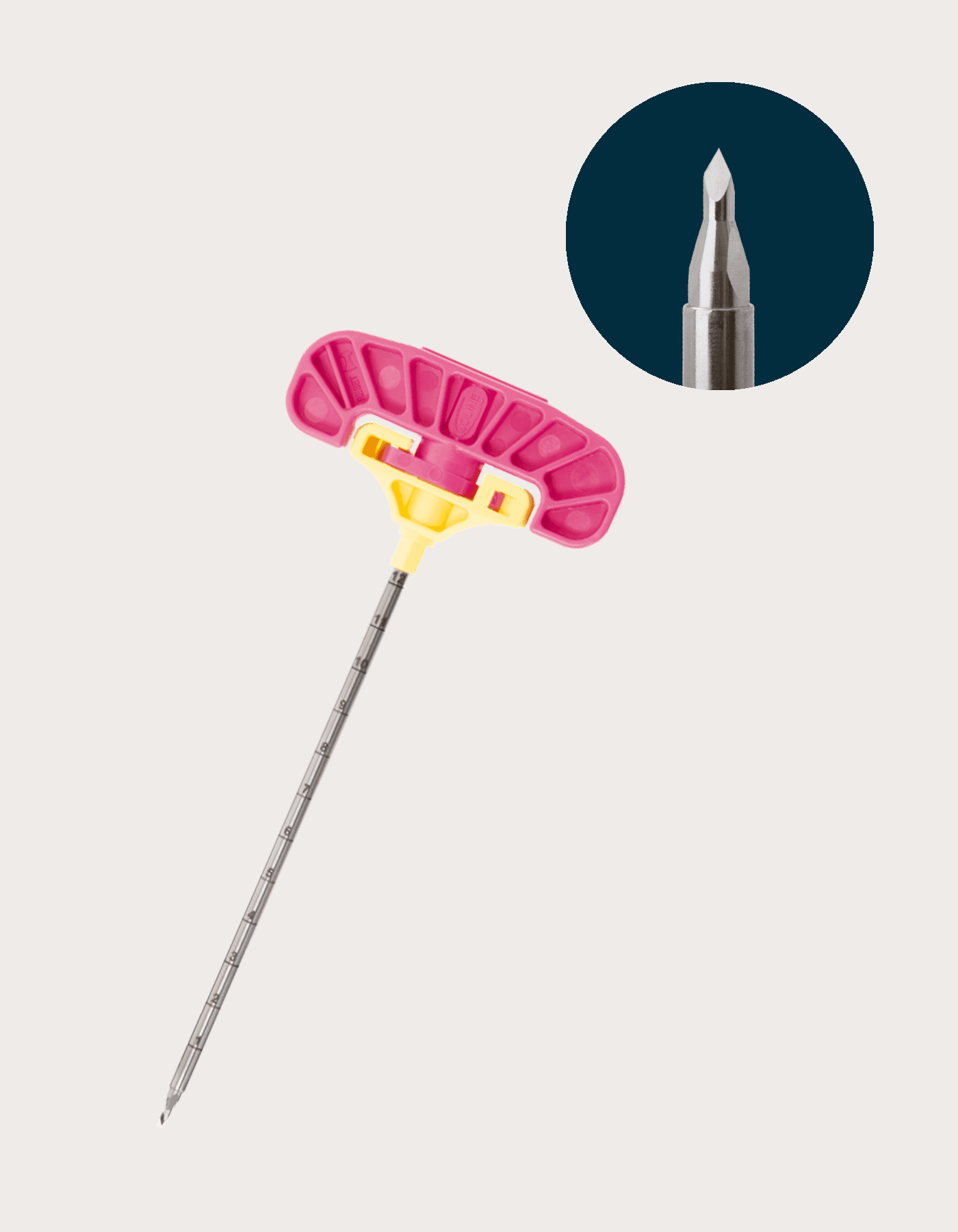
US9312
SpeedTrack Short Tip Introducer Cannula & Stylet

US9203
Introducer Cannula Plug

US9301
Vertebra Biopsy Device Needle & Plunger

US9302
Bone Drill
1
Our kyphoplasty products
Based on many years of experience in catheter technology, Joline can offer a wide range of instruments for the treatment of vertebral compression fractures. Premium quality, high quality materials and functional unique designs of all Joline’s instruments guarantee exceptional medical product quality in compliance with Germany’s high standards.
Special features:
The Joline kyphoplasty system comprises an extensive selection of individual instruments, available separately or as part of a kit.
One-step Device Bone Access Instruments
Hightech-Kypho single- and double-balloon catheters.
Color coding system on Joline access instruments (red, yellow, green) – makes our introducer instruments quick and easy to identify during the procedure.
Joline is your partner in the treatment of vertebral compression fractures

All Joline bone access instruments, balloon catheters, catheter preparation instruments, and application instruments for the treatment of vertebral body fractures are high quality products. They are assembled by hand in an elaborate manufacturing process.
Quality, safety and service are top priorities at Joline.
Not all products are approved in all markets; the approval markings and statements can vary from country to country. Please note the particular country website, which contains additional product-specific information. Products are subject to changes to the design and scope of delivery of the products and further technical development.
